what could you do to change the volume of gas
Gas Laws
The content that follows is the substance of lecture 18. In this lecture we cover the Gas Laws: Charles',Boyle's,Avagadro's and Gay Lussacs as well every bit the Ideal and Combined Gas Laws.
Laws of Gas Backdrop
There are four full general laws that relate the four basic characteristic backdrop of gases to each other. Each constabulary is titled by its discoverer. While information technology is important to understand the relationships covered by each law, knowing the originator is not as important and will be rendered redundant in one case the combined gas law is introduced. And then concentrate on understanding the relationships rather than memorizing the names.
Charles' Law- gives the relationship between volume and temperature if the pressure and the amount of gas are held constant:
1) If the Kelvin temperature of a gas is increased, the book of the gas increases. (P, n Constant)
2) If the Kelvin temperature of a gas is decreased, the volume of the gas decreases. (P, n Constant)
This ways that the volume of a gas is directly proportional to its Kelvin temperature. Recall of it this fashion, if you increase the volume of a gas and must go along the force per unit area constant the only style to achieve this is for the temperature of the gas to increase also.

Calculations using Charles' Law involve the change in either temperature (T2) or volume (V2) from a known starting amount of each (Five1 and Ti):
Boyle's Police force - states that the volume of a given amount of gas held at abiding temperature varies inversely with the practical pressure when the temperature and mass are constant.
The reduction in the volume of the gas means that the molecules are hit the walls more often increasing the pressure, and conversely if the volume increases the distance the molecules must travel to strike the walls increases and they hit the walls less often thus decreasing the pressure.
Similar Charles' Law, Boyle's Law can be used to decide the electric current pressure or book of a gas so long equally the initial states and ane of the changes is known:
Avagadro's Law- Gives the relationship betwixt volume and amount of gas in moles when pressure and temperature are held constant.
If the amount of gas in a container is increased, the volume increases. If the corporeality of gas in a container is decreased, the volume decreases. This is bold of class that the container has expandible walls.
The human relationship is once again directly proportional and then the equation for calculations is
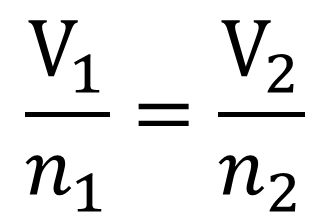
Gay Lussac's Police - states that the force per unit area of a given amount of gas held at constant book is directly proportional to the Kelvin temperature.
If yous heat a gas y'all give the molecules more energy then they move faster. This means more than impacts on the walls of the container and an increase in the pressure. Conversely if yous absurd the molecules downwardly they will ho-hum and the pressure level will be decreased.
To calculate a change in pressure or temperature using Gay Lussac's Police the equation looks like this:
To play around a bit with the relationships, attempt this simulation.
The Ideal Gas Law:
A combination of the laws presented above generates the Ideal Gas Law:
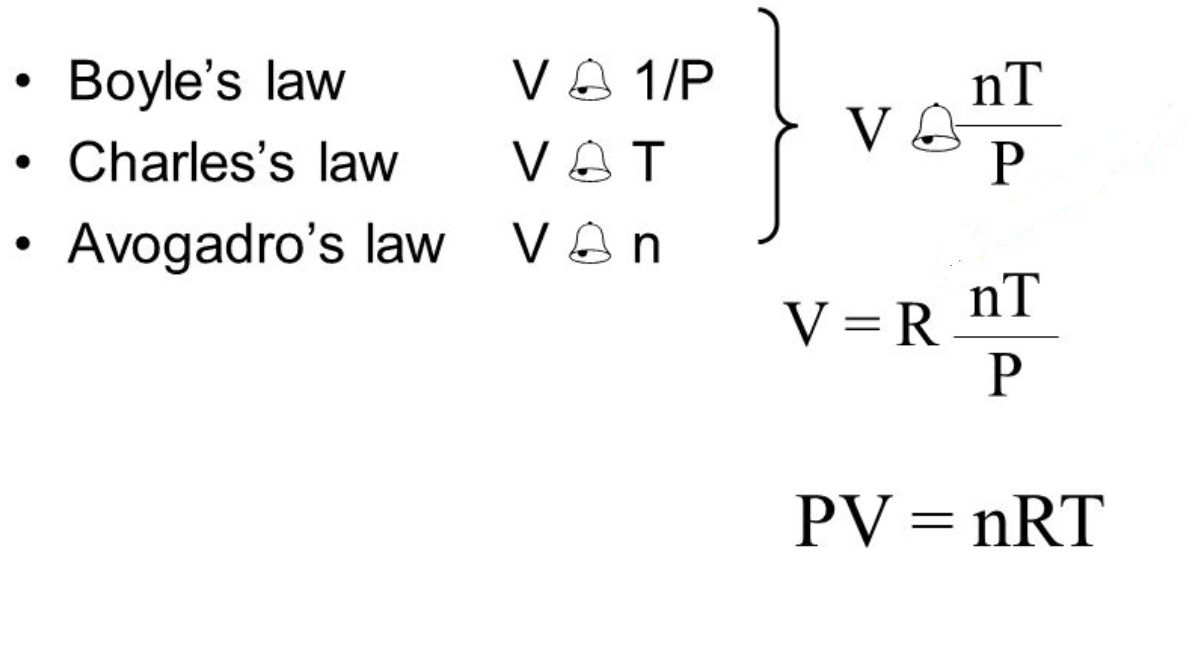
The addition of a proportionality constant called the Ideal or Universal Gas Abiding (R) completes the equation.
As you can see there are a multitude of units possible for the abiding. The only constant about the constant is that the temperature scale in all is KELVIN.
When using the Ideal Gas Law to calculate any property of a gas, you must match the units to the gas constant you choose to apply and you always must place your temperature into Kelvin.
To utilise the equation, you simply need to be able to identify what is missing from the question and rearrange the equation to solve for information technology.
A typical question would be given equally half dozen.2 liters of an ideal gas are contained at 3.0 atm and 37 °C. How many of this moles of the gas are present?
Because the units of the gas constant are given using atmospheres, moles, and Kelvin, it's important to make sure you lot convert values given in other temperature or pressure scales. For this problem, convert °C temperature to Yard using the equation:
T = °C + 273
T = 37 °C + 273
T = 310 G
Now, yous can plug in the values. Solve for the number of moles
due north = PV / RT
n = ( 3.0 atm x 6.2 50 ) / ( 0.08206 L atm /mol K x 310 1000)
northward = 0.75 mol
Hither are some practice problems using the Ideal Gas Police: Practice
The Combined Gas Law
I said higher up that memorizing all of the equations for each of the individual gas laws would get irrelevant after the introduction of the laws that followed. The law I was referring to is the Combined Gas Law:
The combined gas law allows you to derive whatever of the relationships needed past combining all of the changeable peices in the ideal gas law: namely pressure, temperature and volume. R and the number of moles do not announced in the equation as they are generally constant and therefore cancel since they announced in equal amounts on both sides of the equation.
As y'all can meet to a higher place, the equation can be solved for any of the parameters in it. But more than chiefly, you can eliminate from the equation anything that volition remain constant.
For Example, If a question said that a system at 1atm and a volume of ii liters, underwent a alter to 3.5 liters, calculate the new force per unit area, you could simply eliminate temperature from the equation and yield:
P2 = P1Vi/Five2 = (1atm)(2L)/3.5L) = 0.6 atm
Since the question never mentions a temperature we tin assume it remains a abiding and will therefore cancel in the calculation. Y'all should as well think about the answer y'all get in terms of what yous know about the gases and how they act. We increased the book so the pressure should get downwards. Checking our answer, this appears to be right since the pressure level went from 1atm to 0.6atm.
Then the only equation yous really need to know is the combined gas law in order to calculate changes in a gas' properties.
Here are some practice bug with solutions: Practice
Here are some problems for the other gas laws that you lot tin can derive from the combined gas law: Do and Cardinal
collinsimpeartale1949.blogspot.com
Source: https://www.chem.fsu.edu/chemlab/chm1045/gas_laws.html
0 Response to "what could you do to change the volume of gas"
Post a Comment